Water And Water Purification
Introduction
Water is a fundamental compound composed of oxygen and hydrogen, forming the familiar molecule H2O. It can exist in various states, such as vapour, liquid, and solid. Additionally, water may contain dissolved chemicals from the surrounding soil. Pure water is characterized by its clarity, colorlessness, and tasteless properties.
Definition
Safe and wholesome water is defined as water that is:
- Free from pathogenic agents.
- Free from harmful chemical substances.
- Pleasant to the taste, i.e., free from colour and odour.
- Usable for domestic purposes.
Uses of water
- Domestic use: On the domestic front, water is essential for drinking, cooling, washing, bathing, flushing toilets, gardening, and other household activities.
- Public purposes: Water is utilized for cleaning streets, recreational activities such as swimming pools, public fountains, ornamental ponds, fire protection, and maintaining public parks.
- Industrial purposes: Industries, particularly those involved in iron, steel, and paper production, require substantial amounts of water for processing and cooling.
- Agricultural purposes: Irrigation is indispensable for the cultivation of food, fruits, and vegetables.
- Power production: Water is a crucial resource for power production, both through hydropower and steam power.
- Trade and business: Various commercial establishments, including hotels, restaurants, dairies, laundries, and hospitals, depend on an adequate water supply for drinking, bathing, and washing.
Water requirements
- Physiological requirements: The physiological requirement is approximately 2 liters per person.
- Consumption: Water consumption varies based on climate conditions, standards of living, and individual habits.
- Daily supply: An adequate water supply for all urban domestic purposes is considered to be in the range of 150-200 liters per capita per day. For hospitals, a recommended supply is around 45-50 gallons per person per day, while for domestic use in urban areas, it is suggested to be in the range of 20-25 gallons per person per day.
Sources of water
- Rain
- Surface
- Impounding reservoir
- Rivers and streams
- Tanks, ponds and lakes
3. Ground water
- Shallow wells
- Deep wells
- Springs
- Rain

Rain is the primary source of all water. A portion of the rainwater infiltrates the ground, giving rise to groundwater. Another portion evaporates back into the atmosphere, while some runs off to create streams and rivers that eventually flow into the sea. Additionally, water in the soil is absorbed by plants and subsequently evaporated through their leaves.
Characteristics: Rainwater is considered the purest form of water in nature. Physically, it appears clear, bright, and sparkling. Chemically, it is very soft water, containing only minimal traces of dissolved solids. Due to its softness, it can have a corrosive effect on lead pipes. Bacteriologically, rainwater from clean areas is free from pathogenic agents.
Impurities: However, rainwater tends to become impure as it traverses through the atmosphere. It picks up suspended impurities such as dust, soot, microorganisms, and gases like carbon dioxide, nitrogen, oxygen, and ammonia. Gaseous emissions, such as sulphur and nitrogen oxides from power plants using fossil fuels, react with atmospheric water, forming a dilute solution of sulphuric and nitric acid. The precipitation of these acids (acid rain) has begun to exert serious impacts on surface water quality and on plants, among other environmental aspects.
2. Surface water
Surface water originates from rainfall and serves as the primary water source in many regions. Examples of surface water include rivers, tanks, lakes, manmade reservoirs, and seawater. However, surface water is susceptible to contamination from both human and animal sources. It is not safe for human consumption unless it undergoes proper sanitary protection and purification processes before use. Major sources of surface water are:
a. Impounding reservoirs

Artificial lakes, typically constructed with earthwork or masonry, store large quantities of surface water. Dams built across rivers and mountain streams also create significant reservoirs. The area draining into the reservoir is known as the “catchment area.” Storing water for extended periods in reservoirs can lead to the growth of algae and other microscopic organisms, causing unpleasant tastes and odors in the water.
Characteristics: Impounding reservoirs generally provide water of reasonably good quality. The water is typically clear, palatable, and ranks closely to rainwater in terms of purity. If the surrounding hills are covered with peat, the water may acquire a brownish coloration. The water is soft and considered free from pathogenic organisms.
Impurities: Upland surface water gets its impurities from the catchment area, with human habitations and areas for animal keeping and grazing serving as sources. Hence, it is crucial to keep the catchment area free from human or animal intrusion. The common belief that mountain streams are always very pure is often untrue. Even in the absence of human habitation or cattle, there remains a possibility of contamination caused by wild animals.
b. Rivers and streams


The other main sources of water are rivers and streams, which often provide a reliable water supply. However, river water poses a significant challenge as it is consistently heavily polluted and is unsuitable for drinking without proper treatment.
Characteristics: River water tends to be turbid during the rainy season and may appear clear in other seasons. However, the clarity of water does not guarantee its safety for drinking. River water contains various dissolved and suspended impurities, with a potentially high bacterial count, including human intestinal organisms.
Impurities: Rivers are often considered a direct link between the alimentary canals of people living upstream and the mouths of those downstream. The impurities in river water originate from surface runoffs, sewage, sullage water, industrial and trade wastes, and drainage from agricultural areas. Cultural practices such as bathing, animal washing, and disposal of the dead contribute to water pollution.
Purification (self): Some level of self-purification occurs in river water through natural processes such as dilution, sedimentation, aeration, oxidation, sunlight exposure, and the presence of plant and animal life. However, these natural processes are not sufficient to make the water safe for drinking. Therefore, river water requires purification before it can be considered suitable for drinking purposes.
c. Tanks, ponds and lakes


Tanks, ponds, and lakes serve as large excavations for storing surface water, especially in areas without a centralized pipeline water supply. Tanks, in particular, play a crucial role in providing water in some regions. However, these water bodies can face challenges related to impurities and contamination.
Characteristics: Tanks are significant storage reservoirs for surface water, and they are frequently used for various purposes, such as washing clothes, cleaning vehicles, bathing, washing cattle, and even swimming. Older tanks may be overgrown with aquatic vegetation, further impacting water quality.
Impurities: These water bodies are susceptible to contamination by both human and animal activities, including washing, bathing, cleaning vehicles, and swimming. Children defecating around tanks, with subsequent rainwater washing and contamination, poses a high risk for drinking water safety. Additionally, older tanks may harbor aquatic vegetation, and surface drains draining into tanks and ponds contribute to unavoidable contamination.
Natural purification: Similar to river and stream water, some level of natural purification occurs in tanks. However, this natural purification is insufficient to ensure the water is safe for drinking. The presence of impurities, contamination from various activities, and the growth of aquatic vegetation make it necessary to implement additional purification measures before using tank water for drinking purposes.
3. Ground water
Rainwater percolating into the ground forms groundwater, and it is often considered the most cost-effective and practical means of providing water to small communities. Groundwater is generally safer than surface water due to the natural filtering action of the ground. However, it is now recognized that there are limits to groundwater availability worldwide.
Characteristics: Groundwater is likely to be free from bacterial infections, making it generally safe for consumption without the need for extensive treatment. It typically remains available even during dry seasons, providing a reliable water supply. Compared to surface water, groundwater is less prone to contamination. However, it may have high mineral contents such as calcium and magnesium salts, resulting in water hardness. Extracting groundwater usually requires a pump or another method to lift the water to the surface.
It’s important to note that while groundwater is often considered a reliable and safe water source, over-extraction and contamination can still pose challenges. Sustainable management practices are essential to ensure the long-term availability and quality of groundwater.
Sources of ground water.
- Wells
- Tube wells
- Springs
A. Wells
Wells are common sources of water in rural and village areas. They are artificial holes or pits dug deep into the earth to reach the water table, allowing subsoil water to percolate. Various types of wells exist:
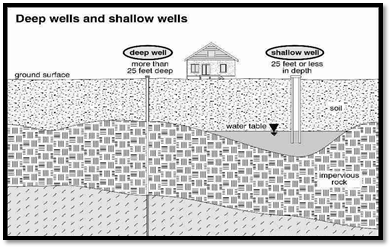
i. According to technical depth:
a. Shallow wells: These wells tap into the topmost subsoil water, located above the first impervious layer in the ground. Shallow wells are susceptible to pollution from nearby sources such as latrines, drains, soakage pits, cesspools, and manure collection.
b. Deep wells: Deep wells tap into water from the water-bearing stratum below the first impervious layer in the ground. Typically machine-dug and reaching several hundred meters in depth, deep wells provide relatively safe drinking water compared to shallow wells. This is because the water in deep wells travels a greater distance through the earth, offering better protection through efficient filtration. The water from deep wells is generally pure and free from bacteria, although inorganic impurities may be present, resulting in hard water. Deep wells ensure a continuous water supply even during dry seasons (summer). Adequate protection of the well’s sides with brickwork set in cement, extending down to the impermeable stratum, prevents the mixing of subsoil water and helps avoid further contamination.
ii. According to methods of construction:
a. Katcha well: A Katcha well is essentially a hole dug into the water-bearing stratum. These wells are more susceptible to contamination and are prone to drying up.
b. Pucca well: A Pucca well is a constructed well made of bricks or stone. While it may dry up earlier than expected, it is less prone to easy contamination compared to Katcha wells.
c. Step well: A Step well is a type of Pucca well where steps are constructed to facilitate access for people to descend into the well for fetching water or quenching their thirst. The presence of steps makes it more convenient for users to access water and contributes to the overall durability of the well structure.

iii. Artesian well
An artesian well is a type of deep well where the water rises above the ground level due to the pressure between two impervious strata. The water in this type of well is tapped through a boring process. Artesian wells share similarities with deep tube wells.

Sources of impurities of wells
- Surface washing can introduce impurities into well water.
- Cracks and fissures in the well can serve as pathways for contaminants.
- Proximity to drains, cesspools, or latrines may lead to the infiltration of pollutants into the well water.
- Trees and plants grown close to the wells can contribute impurities through bird droppings and leaves.
- Impurities may enter the well through vessels and ropes used for drawing water.
- The rise of subsoil water during rains and floods can introduce contaminants into the well.
Improvement of well
The sanitary quality of a well can be improved by implementing the following measures:
- Raising the lining of the well wall: Ensure that the well wall is raised one or two feet above the ground level. This helps prevent surface water runoff and potential contamination from entering the well.
- Covering the top with a concrete slab: Install a concrete slab as a protective cover for the well. This prevents the entry of debris, insects, and other contaminants into the well, safeguarding the water quality.
- Installing one or more hand pumps: Use hand pumps to lift water from the well. This not only provides a convenient means of access but also helps prevent direct contact with the water, reducing the risk of contamination.
- Maintaining proper drainage: Establish a drainage system of at least 10 feet in length around the well. This drainage should be designed to prevent soakage and contamination, ensuring that surface water is directed away from the well.
By implementing these measures, we can enhance the sanitary conditions of the well, promoting the delivery of clean and safe water for consumption.
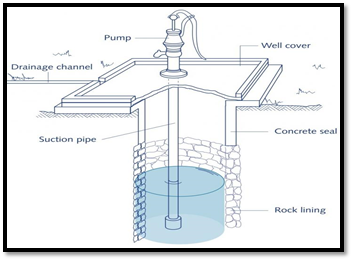
Sanitary well
A sanitary well is one that is constructed properly, located strategically, and protected against contamination to yield a safe water supply. The following important considerations should be taken into account while constructing a sanitary well:
- Location: The well should be tapped in good soil, at least 15 meters (50 feet) away from potential sources of contamination such as latrines, sewage, open drainage, trenching grounds, and other pollution sources. Ideally, it should be situated at least 100-150 feet away from animal pens or sheds. Additionally, the well should be located at a higher elevation to prevent surface contamination. The distance between the well and the users’ houses should be considered to ensure convenient access.
- Lining: The well should be sunk deep into the impervious layer, and its lining should be constructed using bricks or stones in cement, extending up to 6 meters (20 feet) deep to allow water entry from the bottom rather than the sides. The lining should also extend 2-3 feet above the ground level.
- Parapet: A parapet wall should surround the well, reaching a height of at least 70-75 cm above the ground level.
- Platform: The platform should be made of cement concrete, extending at least 3 feet in all directions with a gentle slope outward leading to a drain built along its edge.
- Drain: A durable drain should be constructed to carry off spilled water to a public drain or soakage pit, preventing waterlogging around the well.
- Cover: The top of the well should be securely covered with a cement concrete cover to prevent direct contamination. Covered wells have been proven to provide water that is bacteriologically safe for use.
- Hand pump: A good sanitary well should be equipped with a hand pump to lift water. Studies indicate that wells with fitted pumps exhibit better water quality from a bacteriological standpoint. The use of a hand pump reduces the risk of contamination compared to people using their own buckets and ropes.
- Public responsibility: While a sanitary well is crucial, it does not guarantee freedom from waterborne diseases unless consumers take precautions at the individual and family levels. People should be educated and motivated to maintain strict cleanliness around the well, avoiding activities such as washing clothes, cleaning animals and humans, and dumping refuse. The use of a common bucket and rope for water extraction is recommended to prevent contamination. Health education plays a vital role in achieving this.
- Quality: The water’s physical, chemical, and bacteriological quality should conform to acceptable standards to ensure it is safe and wholesome.
- Amount of water: The depth and size of the well can vary. To estimate the quantity of water in a well, the following formula can be used:
D2xWx5= Number of gallons of water in a well, where
D= Diameter of a well (feet)
W= Depth of water (feet)
E.g.- A well is 5 Ft. diameter which has 20 Ft. of water depth. Now the quantity of water inside the well will be:
Quantity of water= D2xWx5
=52x20x5
= 2500 gallons of water
If the nurse is chlorinating the well for purification, she can use this volume to calculate the amount of chlorine needed based on the required dosage per gallon.
b. Tube Wells: Tube wells can be classified into shallow tube wells and deep tube wells. This system is commonly employed in Terai and hilly regions like Kathmandu, offering a bacteriologically safe and cost-effective water source.
i. Shallow Tube Wells: Shallow tube wells are suitable when the water is located close to the ground surface, typically within a depth of 18-36 feet. They can be bored into gravel, coarse sand, or chalk, but clay soils or fine sand are not suitable. Shallow tube wells find utility in villages, isolated houses, camps, and during festivals.
ii. Deep Tube Wells: Deep tube wells involve drilling through successive substrata of gravel or rock until an adequate supply of groundwater is reached. These wells can be several hundred feet deep, requiring sophisticated mechanical drilling equipment and skilled engineering expertise. Although costly to construct and operate, deep tube wells are considered an ideal source for providing a protected water supply.
c. Springs: Springs occur when groundwater naturally emerges and flows under pressure at the surface. Springs can be categorized into shallow springs and deep springs. Shallow springs tend to dry up quickly during summer months, whereas deep springs exhibit more consistent water flow throughout the seasons. In certain geographic areas, springs play a crucial role as a water source. Exploiting springs is simpler than other sources as there is no need for pumping to bring water to the surface. However, springs are susceptible to contamination, emphasizing the importance of well-built protective structures to ensure the quality of the water.

Water-related diseases
The health of individuals can be adversely impacted by the consumption of contaminated water, either directly or through food, as well as by the use of contaminated water for personal hygiene and recreation. The term “water-related diseases” encompasses classical waterborne diseases, with developing countries bearing a significant burden, the most severe being diarrheal diseases. The categorization of water-related diseases is as follows:
a. Biological (Waterborne diseases)
- Viral: Hepatitis A, Hepatitis E, poliomyelitis, rotavirus diarrhea in infants
- Bacterial: Typhoid, bacillary dysentery, E. coli diarrhea, cholera
- Protozoal: Amoebiasis, giardiasis
- Helminthic: Roundworm, threadworm
- Vector-Borne: Snail: schistosomiasis
- Cyclops: Guinea worm, fish tapeworm
b. Chemical
Chemical pollutants of various types derived from industrial and agricultural wastes are increasingly infiltrating public water supplies. These pollutants encompass detergent solvents, cyanides, heavy metals, minerals, organic acids, nitrogenous substances, bleaching agents, dyes, pigments, sulphides, ammonia, toxic and biocidal organic compounds of great variety. These pollutants may affect human health not only directly but also indirectly by accumulating in aquatic life, such as fish, which is used as human food. The current concern about chemical pollutants in water relates not only to their acute toxic effects on human health but also to the potential long-term effects of low-level exposure, which are often nonspecific and difficult to detect.
In addition to the above, water is associated with the following health conditions:
- Dental health: Presence of fluoride at about 1 mg/lit in drinking water is known to protect against dental caries, but high levels of fluoride cause mottling of dental enamel.
- Cyanosis in infants: High nitrate content of water is associated with methemoglobinemia. This is a rare occurrence but may happen when surface water from farmland, treated with fertilizer, gains access to the water supply.
- Diseases related to disease-carrying insects breeding in or near water: Malaria, filaria, etc.
- Diseases transmitted due to inadequate water use: Shigellosis, trachoma and conjunctivitis, ascariasis, scabies
While pollution appears to be an inevitable consequence of modern industrial technology, the current challenge is to determine the level of pollution that allows for economic and social development without presenting hazards to health.
Purification of water
The primary aim of water purification is to safeguard the water supply by eliminating organic matter, suspended particles, and dissolved substances present in water. The purification process aims to ensure the delivery of clean and safe water. Two main methods are employed for purifying impure water
- Natural methods
- Artificial methods
- Natural methods
The natural methods can be any of the following:
- Evaporation and condensation (conversion of vapour to liquid.)
- Storage and settlement
- Air, sunlight and oxidation
- Filtration through earth
- Biological purification
- Artificial methods
The primary purpose of water treatment is to generate water that is safe and wholesome, significantly contributing to human health. The method of treatment chosen depends on the characteristics of the raw water and the desired standards of water quality. Water treatment is conducted on two scales:
- Purification of water on a large scale
- Purification of water on a small scale
- Purification of water on a large scale
The primary objective of water treatment is to produce water that is safe and wholesome for consumption. The selection of treatment methods depends on the characteristics of the raw water and the desired standards of water quality. For instance, groundwater sourced from wells and springs may require minimal treatment, often limited to disinfection processes. On the other hand, surface water, such as river water, which is prone to turbidity and pollution, necessitates more comprehensive treatment. Components of a typical water purification system may include one or more of the following measures:
- Storage
- Filtration
- Disinfection

- Storage
Water is extracted from its source and collected in natural or artificial reservoirs, creating a water reserve that effectively prevents further pollution. Storage facilitates a substantial amount of natural purification, which can be examined from three perspectives:
a. Physical: Approximately 90% of suspended impurities settle within 24 hours through gravity, resulting in clearer water. This improved clarity allows for better light penetration and reduces the workload on filters.
b. Chemical: Certain chemical transformations occur during storage. Aerobic bacteria oxidize the organic matter in the water with the assistance of dissolved oxygen. Consequently, the content of free ammonia decreases, and there is an increase in nitrates.
c. Biological: During storage, the bacterial count decreases, and pathogenic organisms gradually perish. It has been observed that when river water is stored, the total bacterial count can decrease by as much as 90% within the initial 5-7 days. This stands as one of the significant advantages of storage. The optimal storage period for river water is considered to be around 10-14 days. Prolonged storage may lead to the potential development of vegetation, such as algae, contributing to unpleasant odours and discoloration in the water.
II. Filtration
Filtration constitutes the second pivotal stage in water purification, playing a crucial role in removing approximately 98-99% of bacteria, in addition to other impurities. Two primary types of filters are employed: “biological or slow sand filters” and “rapid sand or mechanical filters.”
a. Slow sand or biological filters:
In this method, water is drawn from rivers, streams, or lakes into filter beds, comprised of water-tight rectangular basins typically constructed with masonry. Each unit must have dimensions of at least 50’x40’x15 at the bottom, covering an acre (0.405 hectares) or more. Multiple units are necessary for continuous filtration without plant shutdowns. These filter beds are usually situated in the outer districts of a city or town, positioned along the banks of rivers or streams. Before reaching the filter tanks, water undergoes an initial stage in setting tanks or large artificially constructed reservoirs, facilitating sedimentation and eliminating a significant number of bacteria.
Filter bed construction:
- A coarse gravel layer of bricks or broken stones at the bottom, with a thickness of 6 inches to 12 inches.
- Above the coarse gravel layer, another layer of gravel, 12 inches deep, is added.
- Above the sand, 4-5 feet of water is allowed to stand for filtration.
- Perforated tubular drains at the bottom of the bed collect the filtered water.
Advantages of slow sand filters:
- Simple construction and operation.
- Lower construction costs compared to rapid sand filters.
- High-quality filtered water, with a bacteriological quality of 99.9 to 99.99% pathogen-free.
- Eliminates the need for chemical coagulation before filtration.
Disadvantages:
- Requires a large area.
- Sedimentation must precede the filtration process.
- Filtration rate is slow, typically at 2 gallons per square foot per hour or 50 gallons per square foot per day.
- Less operationally feasible than sand filters.
b. Rapid sand or biological filtration:
Rapid sand filters, as the name suggests, operate at a speed that is 100 times faster than slow sand filters. They are classified into two types:
A. Gravity type (Paterson’s filter)
B. Pressure type (Candy’s filter).
A. Gravity type: The gravity type comprises a unit of filter bed with a surface area of 90 sq. ft. It includes under drains with perforated pipes covered by a layer of graded gravel, upon which a layer of coarse sand is laid. In this rapid filtration type, the following steps are involved:
i. Coagulation: In the first step, raw water collected from rivers or lakes is treated with a chemical coagulant such as alum or aluminum sulfate. The dosage varies from 5-40 mg or more per liter based on the turbidity, color, temperature, and pH of the water.
ii. Mixing: The treated water is then passed into the ‘mixing chamber,’ causing violent agitation for a few minutes to ensure thorough mixing of the chemicals in the water.
iii. Flocculation: The next stage involves gentle and slow stirring of the treated water in a flocculation chamber for about 30 minutes. During this time, a flocculent material, aluminum hydroxide, is formed due to the interaction of alum with the calcium carbonate present in raw water. The white precipitate settles at the base as aluminum hydroxide.
iv. Sedimentation: The coagulated water is directed into sedimentation tanks for a period of 2-6 hours. During this time, the precipitate and impurities, including bacteria, settle down in the tank. About 95% of the precipitate is removed before proceeding to rapid sand filtration.
v. Filtration: The filter bed, with a surface area of about 80 to 90 square meters, consists of three layers: a 1-meter sand bed, a layer of graded gravel (30-40 cm) supporting the sand bed, and a water depth of 1-1.5 meters. As filtration progresses, the alum floc not removed by sedimentation is held back on the sand bed, forming a slimy layer similar to the zoogleal layer in slow sand filters. This layer absorbs bacteria and facilitates purification by oxidizing ammonia. During filtration, impurities and bacteria may clog the filter, necessitating periodic washing or “backwashing.” This involves reversing the water flow through the sand bed to dislodge impurities and clean the bed. Backwashing may also involve using compressed air.
Advantages
- Can directly handle raw water without requiring storage.
- Filtration is significantly faster, around 40-50 times faster than slow filters.
- Washing of the filter is easier.
- Offers more flexibility in operation.
- Requires less space occupation.
B. Pressure type (Candy’s filter): The pressure type filter is noteworthy, especially for smaller installations such as swimming pools and commercial facilities where turbid water requires cleaning and rendering fit for use, particularly in boilers. This system operates under a pressure higher than atmospheric pressure. The filter bed maintains the same thickness, and the size of sand and gravel is consistent with that of the gravity type. The primary distinction lies in the fact that, in the pressure type, raw water treated with a coagulant parse onto the top of the sand and forms a gelatinous film without undergoing previous mixing, flocculation, or settlement.
Compared to the gravity type, pressure filters tend to get clogged more rapidly and require more frequent cleaning. Despite this drawback, a significant advantage is that pressure filters can be installed on a rising main without the need for additional pumping.
III. Disinfection: It is one of the most widely used disinfectants for effectively eliminating pathogenic organisms. However, it should be noted that chlorine has no impact on spores and certain viruses, such as polio and hepatitis. It is crucial to emphasize that chlorine serves as a supplement rather than a substitute for sand filters. As an oxidizing agent, chlorine plays a multifaceted role it oxidizes iron, manganese, and hydrogen sulfide, while also acting to destroy certain taste and odor-producing constituents. Additionally, chlorine helps control algae and slime organisms, and it aids in the coagulation process. Chlorine can be applied in various forms, including gas, solution, bleaching powder, or chlorine tablets.
Principles of chlorination
- Water intended for chlorination should be clear and free from turbidity. Clear water ensures that chlorine can effectively reach and act on the target microorganisms.
- It is crucial to estimate the chlorine demand of the water to avoid over-dosing. Over-dosing can lead to an excess of chlorine, which may result in undesirable by-products and potential health risks.
- A minimum contact period of at least 30 minutes is essential to ensure the effective killing of bacteria and viruses. Sufficient contact time allows chlorine to interact with and neutralize harmful microorganisms in the water.
- The recommended concentration of free chlorine should not fall below 0.5 mg/hour. This minimum level is necessary to maintain effective disinfection, particularly in cases where the water is highly polluted. It is important to account for chlorine demand when determining the appropriate concentration to achieve desired disinfection levels.

- Purification of water on small scale
I. Household purification of water
- Boiling
- Chemical disinfection
- Filtration
- SODIS (Solar Water Disinfection)
II. Disinfection of wells
- Boiling: Boiling is a satisfactory method for purifying household water. To be effective, water must reach a “rolling boil” for 5-10 minutes, eliminating all bacteria, spores, cysts, and ova. Boiling also reduces temporary water hardness by expelling carbon dioxide and precipitating calcium carbonate. Though the taste may change, it is harmless. While boiling is an excellent purification method, it provides no “residual protection” against subsequent microbial contamination. It is advisable to boil water in the same container intended for storage to prevent contamination during storage. Careful handling is essential to avoid recontamination. Clean hands are crucial, and no dirty vessels should be immersed in the container when collecting water. This is imperative for community health in preventing waterborne diseases.

- Chemical disinfection
- Bleaching powder: – Bleaching powder, also known as chlorinated lime (CaOCl2), is a white, shapeless powder with a distinct chlorine odour. When freshly produced, it contains approximately 33% available chlorine. However, it is an unstable compound that rapidly loses its chlorine content when exposed to air, light, and moisture. Therefore, it should be stored in a dark, cool, dry place in a closed container resistant to corrosion. Regular monitoring of the chlorine content in bleaching powder stocks is essential. Approximately 1 ounce (about 28 gm.) of bleaching powder is needed to disinfect 1000 gallons of water. It is important to note that highly polluted and turbid waters are not suitable for direct chlorination.
2. Chlorine solution: – Chlorine solution can be prepared from bleaching powder. If 4 kg of bleaching powder with 25% available chlorine is mixed with 20 liters of water, it will yield a 5% chlorine solution. Alternatively, ready-made chlorine solutions in various strengths are available in the market. Similar to bleaching powder, chlorine solutions are susceptible to losses when exposed to light or stored for extended periods.
To maintain its effectiveness, the chlorine solution should be stored in a dark, cool, and dry place within a closed container. Regular monitoring and replacement, as needed, are crucial to ensure the solution retains its desired concentration for effective water disinfection.
3. High test Hypochlorite (HTH): – High Test Hypochlorite, also known as perchloron, is a calcium compound containing 60-70% available chlorine. It exhibits greater stability compared to bleaching powder and experiences minimal deterioration during storage. Solutions derived from HTH are commonly employed for water disinfection purposes.
4. Chlorine tablets: – Chlorine tablets, sold under various trade names such as halazone tablets, are readily available in the market. They are highly effective for disinfecting small quantities of water. These tablets come in various strengths and are now abundant in the market at an affordable price. Typically, a single tablet weighing 0.5 grams is adequate to disinfect 20 liters of water.
5. Iodine: Iodine can be employed for emergency water disinfection. Two drops of a 2% ethanol solution of iodine are sufficient for one liter of clear water. An effective disinfection requires a contact time of 20-30 minutes. Iodine remains in its active molecular form over a broad range of pH values and water conditions and persists longer than both chlorine and bromine. However, its major disadvantages include high costs and physiological activity, particularly thyroid activity.
6. Potassium permanganate: Potassium permanganate, an oxidizing agent, has been historically used for water disinfection. Despite its potent oxidizing properties, it is not recommended for water disinfection due to various drawbacks. The suggested dose is 0.5 gm/liter of water. While it may kill cholera vibrios, its efficacy against other disease organisms is limited. Furthermore, potassium permanganate alters the color, smell, and taste of water.
7. Piyush or water guard: For water disinfection, a 0.5% chlorine solution prepared as Piyush can be used by adding 3 drops to 1 liter of water. Shake well and consume after 30 minutes. Water Guard can be utilized by adding 2 ml to 10 liters of water. Stir well with a clean utensil and consume after 30 minutes. These methods are user-friendly and also contribute to fuel savings.

C. Filtration: –
It removes 98-99% of bacteria. Water can be purified on a small scale by filtering through:
- Slow sand filter
- Charcoal filter
- Bio-sand filter
b. Mechanical or rapid sand (domestic) filter
- Slow sand filter: A slow sand filter is a household adaptation of a sand filter. The process is simple – users pour water into the top of the filter and collect the filtered water from the outlet pipe into a bucket. However, over time, especially when dealing with turbid source water, the flow rate may decrease.
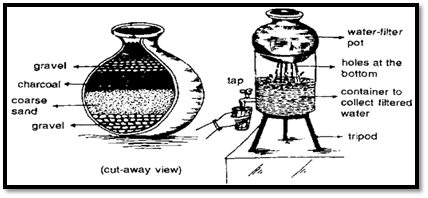
i. Charcoal filter: In this system, three pots are required. Begin by taking one pot and placing a mixture of pebbles, sand, and charcoal to create a filter. Set this filter on top of the second receptacle, making sure to create a hole at the bottom of the first receptacle. To use the system, pour water into the top filter. The water will pass through the layers of pebbles, sand, and charcoal, and the purified water can then be collected from the bottom receptacle, providing a reliable method for obtaining drinking water.
ii. Bio-sand filter (Kanchan arsenic filter): This filter was developed by researchers at the Massachusetts Institute of Technology, the Environment and Public Health Organization of Nepal, and the Rural Water Supply and Sanitation Support Program of Nepal. This innovative household drinking water treatment device is designed to remove arsenic, pathogens, iron, turbidity, odor, and some other contaminants from drinking water.

b. Mechanical or domestic filter: In this type of filter, water is purified using a filtered candle. The principle behind these filters is that water is directed through porous candles, which can be coarse or fine-grained depending on the material they are made of. While they effectively filter out bacteria, they may not be as effective against viruses. Filter candles are prone to clogging with impurities and bacteria, so it is recommended to clean them by scrubbing with a hard brush under running water and to boil them at least once a week. It is crucial to use only clean water with ceramic filters.
There are two common types of filters used for domestic purposes:
- Barkefeld filter: This filter consists of a candle made of porcelain. The filter setup includes two drums, one positioned over the other. A porous filter is fixed to the bottom of the upper drum, where impure water is stored. Pure water then flows into the lower drum.

- Water purifiers: This process involves the removal of undesirable chemicals, materials, and biological contaminants from contaminated water. It effectively eliminates germs through the use of ultraviolet rays, resulting in obtaining germ-free water from water purifiers such as Uroguard.


SODIS (Solar Water Disinfection): The SODIS method is ideal for treating water for drinking in developing countries. The UV-A rays in sunlight play a crucial role in killing germs, including viruses, bacteria, and parasites. SODIS is an inexpensive and simple water purification method that utilizes sunlight and drink bottles. It only requires sunlight, empty plastic soft bottles, and preferably a black surface. This method is widely adopted on a household scale. In Nepal, SODIS has been implemented since 2000.
Process of SODIS
- Take 1-2 liters of a transparent plastic bottle and remove the label. A large bottle is not useful because UV rays can penetrate only up to 10 cm of the water level. The bottle should be clean and not crushed.
- Fill the bottle halfway with clean water.
- Cover the bottle with its cap and shake it up and down for 20 times. This increases the amount of oxygen inside the bottle, which helps kill microorganisms and improves the taste. This water should not be discarded.
- After shaking, fill the bottle with the full volume of water.
- Place the bottle horizontally on a flat surface in direct sunlight from sunrise to sunset for 8-9 hours to ensure maximum exposure to sunlight. The process is more effective if the bottom is painted black or placed on a black sheet of plastic that absorbs more heat, aiding in killing more microbes.
- If there is any cloudiness, the bottle should be kept for 2 days. Now, the water is safe and suitable for drinking.
Advantages
- Proven reduction of viruses, bacteria, and protozoa in water.
- Simplicity of use, inexpensive, and widely accepted.
- No cost if using recycled plastic bottles.
- Minimal change in the taste of the water.
- Recontamination is low because water is served and stored in small, narrow-necked bottles.
Disadvantages
- Limited volume of water that can be treated at once.
- This process is not effective without sunlight.
- Length of time required to treat water.
- A large supply of intact, clean, suitable plastic bottles is required.
- It reduces the number of microorganisms but does not chemically purify contaminated water.

II. Disinfection of wells
Disinfection of wells is crucial in rural areas where they serve as the primary water supply, as they can be prone to contamination leading to waterborne diseases like cholera and gastroenteritis. The cost-effective method for well disinfection involves using bleaching powder. Here are the corrected steps for well disinfection:
- Determine the well water volume:
- Measure the depth of water (h) in meters.
- Measure the diameter of the well (d) in meters.
- Use the formula:
Volume (liter) = 3.14xd2xhX1000/4
- Calculate bleaching powder quantity: Approximately 2.4 grams of high-quality bleaching powder is required to disinfect 1000 liters of water. This results in an approximate dose of 0.7 mg of chlorine per liter of water.
- Dissolve bleaching powder: Place the required bleaching powder in a bucket with coarse sand (up to 100 gm. per bucket). Allow the mixture to settle for 5-10 minutes, letting the lime settle down.
- Deliver chlorine solution into the well: Lower the bucket containing the chlorine solution below the water surface. Mix the water vigorously by moving the bucket vertically and laterally.
- Contact period: Allow a contact period of one hour before drawing water for use.
- Orthotolidinearsenite test: Perform the test to check for residual chlorine at the end of the one-hour contact. If the free residual chlorine is less than 0.5 mg/liter, repeat the chlorination procedure.
- Optimal disinfection timing: Wells are best disinfected at night, following the day’s water draw-off.
Ensure that these steps are followed carefully to effectively disinfect wells and prevent waterborne diseases in rural areas.
Hardness of Water
Hardness can be defined as the soap-destroying property of water. Water is considered hard when large amounts of soap are needed to produce lather. The hardness in water is primarily caused by four dissolved compounds, which are:
- Calcium bicarbonate
- Magnesium bicarbonate
- Calcium sulfate
- Magnesium sulfate
The presence of any one of these compounds causes hardness. There are other compounds of lesser importance, such as calcium and magnesium chlorides and nitrates, which can also contribute to hardness but typically occur in small amounts. Iron, manganese, and aluminum compounds can also cause hardness; however, since they are usually present in minimal quantities, they are not commonly considered when discussing water hardness.
Hardness is classified as carbonate and non-carbonate.
- Carbonate hardness which was formerly designated as “temporary” hardness is due to the presence of calcium and magnesium bicarbonates.
- Non-carbonate hardness formerly designated as “permanent” hardness, is due to calcium and magnesium sulphates, chlorides and nitrates.
Hardness in water is expressed in terms of “milli equivalents per litre (mEq/L)”. One mEq/L of hardness producing ion is equal to 50 mg CaC0 (50ppm) in one litre of water. The terms soft and hard water are used when the levels of hardness are as given below.
Classification of hardness in water
Classification Level of hardness (in Eq/Liter)
a. Soft water less than 1 (<50 mg/L)
b. Moderately hard 1-3 (50-150 mg/L))
c. Hard water 3-6 (150-300mg/L)
d. Very hard water Over 6 (> 300mg/L)
Drinking water should be moderately hard. Softening of water is recommended when the hardness exceeds 3 mEq/l (150 mg per liter).
Advantages of hardness
- Reduced prevalence of arteriosclerotic heart disease
- Lower rates of degenerative heart disease
- Decreased risk of sudden death due to cardiac arrest
- Possible reduction in hypertension
- Prevention of tetany (due to the higher calcium and magnesium content)
Disadvantages of hardness
Hardness in water presents several disadvantages both to the domestic and industrial consumer. These may be stated as follows:
- hardness in water consumes more soap and detergents
- when hard water is heated, the carbonates are precipitated and bring about furring or scaling of boilers. This leads to great fuel consumption, loss of efficiency and may sometimes cause boiler explosions
- hard water adversely affects cooking; food cooked in soft water retains its natural colour and appearance
- fabrics washed with soap in hard water do not have a long life
- there are many industrial processes in which hard water is unsuited and gives rise to economic losses
- hardness shortens the life of pipes and fixtures.
Special treatment
(a) Removal of hardness
The methods of removal of hardness are briefly stated as below:
Temporary hardness
- Boiling
- Addition of lime
- Addition of sodium carbonate
- Permutit process.
Permanent hardness
- Addition of sodium carbonate
- Base exchange process.
1. Boiling
Boiling removes the temporary hardness by expelling carbon dioxide, and precipitating the insoluble calcium carbonate. It is an expensive method to soften water on a large scale.
2. Addition of lime
Lime softening not only reduces total hardness but also accomplishes magnesium reduction. Lime absorbs the carbon dioxide, and precipitates the insoluble calcium carbonate. In the Clark’s method of softening water, one ounce of quick lime is added to every 700 gallons of water for each degree (14.25 ppm.) of hardness.
3. Addition of sodium carbonate
Sodium carbonate (soda ash) removes both temporary and permanent hardness.
4. Base exchange process
In the treatment of large water supplies, the permutit process is used. Sodium permutit is a complex compound of sodium, aluminum and silica. It has the property of exchanging the sodium cation for the calcium and magnesium ions in the water. When hard water is passed through the permutit the calcium and magnesium ions are entirely removed by base exchange and the sodium permutit is finally converted into calcium and magnesium permutit. By this process, water can be softened to zero hardness. Since water of zero hardness is corrosive, a part of the raw water is mixed with the softened water to secure the desired hardness. After permutit has been used for sometime, it loses its effectiveness but it may be regenerated by treating with concentrated solution of sodium chloride or brine and washing away the soluble calcium and magnesium · chloride formed. Permutit process removes both temporary and permanent hardness.
Water hardness and Cardiovascular Diseases
Studies from various countries have shown a potential inverse relationship between water hardness and cardiovascular disease mortality. Areas with soft drinking water exhibit higher rates of arteriosclerotic heart disease, degenerative heart disease, hypertension, and sudden cardiovascular deaths. However, this link is based on statistical associations and circumstantial evidence. Further research is ongoing to determine a more definitive connection between water characteristics and cardiovascular health.
(b) Fluoridation of water
Fluorine is one of the constituents naturally present in water supplies. In fact, the main source of fluorine is drinking water. Deficiency of fluorine in drinking water is associated with dental caries, and excess with dental and skeletal fluorosis. The term “fluoridation” has been given to the process of supplementing the natural fluoride content of potable waters to the point of optimum concentration. The WHO in 1969 recommended fluoridation of community water supplies in areas where the total intake of fluorides by the population is below the optimal levels for protection against dental caries. Fluoridation is now an accepted public health procedure in many developed countries.
(c) Defluoridation
In some geographic areas, water may contain a high level of fluorides. In such communities, water is defluoridated by phosphate to reduce fluorides to optimum levels.
Sources: Park, K. (2021). Park’s Textbook of Preventive and Social Medicine (26th ed.). Bhanot Publishers.
More details: Click here
For additional link: Click here